Chromium ii nitrate formula4/5/2023 
Washing soda is a hydrate of sodium carbonate. However, when additional aqueous hydroxide is added the When aqueous sodium hydroxide is added to a solution containing lead(II) nitrate, a solid precipitate forms. 99.Write the balanced chemical equation for each of these reactions. Dewater the sludge and dispose of in singlepurpose dumps.ĬhroMiuM(III) nitrate nonahydrate, 99% 500GR ChroMiuM nitrate nonahydrate, 99.99%, (trace Metal basis) CHROMIUM(III) NITRATE NONAHYDRATE FOR AN ChroMiuM(III) nitrate nonahydrate >=99.99% trace Metals basis Chromium nitate Chromium(III) nitrate nonahydrate, 98.5% trace metals basis Chromium(III) Nitrate Nonahydrate98.0-101.0% Chromium isotopic standard Chromium(III) nitrate nonahydrate, nonahydrate Chromium(III)nitrate,nonahydrate(1:3:9) Chromiumtrinitratenonahydrate Nitricacid,chromium(3+)salt,nonahydrate CHROMIC NITRATE NONAHYDRATE CHROMIC NITRATE 9H2O CHROMIUM NITRATE, NONAHYDRATE CHROMIUM NITRATE, HYDROUS CHROMIUM NITRATE CHROMIUM(III) NITRATE NONAHYDRATE CHROMIUM (III) NITRATE ENNEAHYDRATE CHROMIUM(III) NITRATE-9-HYDRATE CHROMIUM (IC) NITRATE CHROMIUM (IC) NITRATE NONAHYDRATE CHROMIUM(III) NITRATE-9-HYDRATE PURE CHROMIUM(III)-NITRATE SOLUTION 65 % CR(NO3)3 9 H2O CHROMIUM(III) NITRATE NONAHYDRATE 99% CHROMIUM(III) NITRATE NONAHYDRATE, 99.99 +% Chromiun Nitrate Nonahydrate CHROMIUM NITRATE (NONAHYDRATE) (CHROMIUM NITARTE) Chromium(III)nitratenonahydrate,98+% Chromium(III)nitratenonahydrate,99.99%min(metalsbasis) CHROMIUM NITRATE, NONAHYDRATE 99.998% CHROMIUM NITRATE, NONAHYDRATE REAGENT ChromiumNitrateSolution Chromium(III)nitratenonahydrate,99% CHROMIUM(III)NITRATE,HEXAHYDRATE Chromium(III) nitrate nonahydrate, 98.5% ChroMiuM nitrate nonahydrate, (trace Metal basis) ChroMiuM(III) nitrate nonahydrate, 99% 100GR CHROMIUM NITRATE, NONAHYDRATE, 99.99%CHROMIUM NITRATE, NONAHYDRATE, 99.99%CHROMIUM NITRATE, NONAHYDRATE, 99.99%CHROMIUM NITRATE, NONAHYDRATE, 99.99% CHROMIUM NITRATE, NONAHYDRATE, REAGENTCHROMIUM NITRATE, NONAHYDRATE, REAGENTCHROMIUM NITRATE, NONAHYDRATE, REAGENTCHROMIUM NITRATE, NONAHYDRATE, REAGENT HighPurS - Chromium(III) Nitrate (Cr(NO3)2*9H2O)- min. Violent reaction with many compounds, including reducing agents alcohols, chemically active metals combustible materials, strong acids, alkaline earth sulfides, aluminum carbides, aluminum, amines, calcium sulfide, carbides, chlorine trifluoride, glycerin, hydrides, hydrochloric acid, hydrogen peroxide, hydrogen sulfide, hydroxylamine, magnesium, metal powders, metal sulfides, molybdenum, phenylhydrazine, phosphorous red/friction, phosphorous trichloride, silicon, sulfides, sulfur, sulfur Chromium Nitrate 845 dioxide, sulfur/friction, sulfuric acid, tungsten, hydrogen trisulfide Waste Disposal Contact with reducing agents fuels, ethers, and other flammable and combustible materials cause a fire and explosion hazard. An aqueous solution of this salt becomes green on heating but reverts to the violet colour on cooling. Evaporation of the methanolic solution in high vacuum over CaCl2 eventually yields pure deep violet rhombic crystals of chromium (III) nitrate nonahydrate. When this solution is diluted to 130,000ppm of the salt, analysis detected by ICP/MS gave the following trace elements (ppm in brackets): Sr (33), Na (3.5), Mo, Ni, Cu, Si and Mg (0.13 each), Se, Zn, Al (0.026 each), Ti (1.5), Fe (4.9), Co (0.07), Sn (0.065), Ba (0.013 and W (0.078). A bluish colour develops as Cr(VI) is reduced to Cr(III). A solution is prepared by dissolving 1g of CrO3 in 3mL of H2O and 2mL of pure HNO3, and carefully, with stirring (behind a screen) pure MeOH (0.5-1.0mL) is added dropwise carefully (shield) with cooling to avoid a violent reaction. The pure deep violet nonahydrate salt is best prepared freshly from pure recrystallised (3 times) chromium (VI) trioxide and pure nitric acid. UN2720 Chromium nitrate, Hazard Class: 5.1 Labels: 5.1-Oxidizer Purification Methods Potential ExposureĬhromium nitrate is used in the preparation of chrome catalysts, in textile printing operations and as a corrosion inhibitor. Chromium(III) nitrate as a source of chromium is added to food supplements for nutritional purposes. 
It is used in the preparation of chrome catalysts, in textile printing operations, and as a corrosion inhibitor. 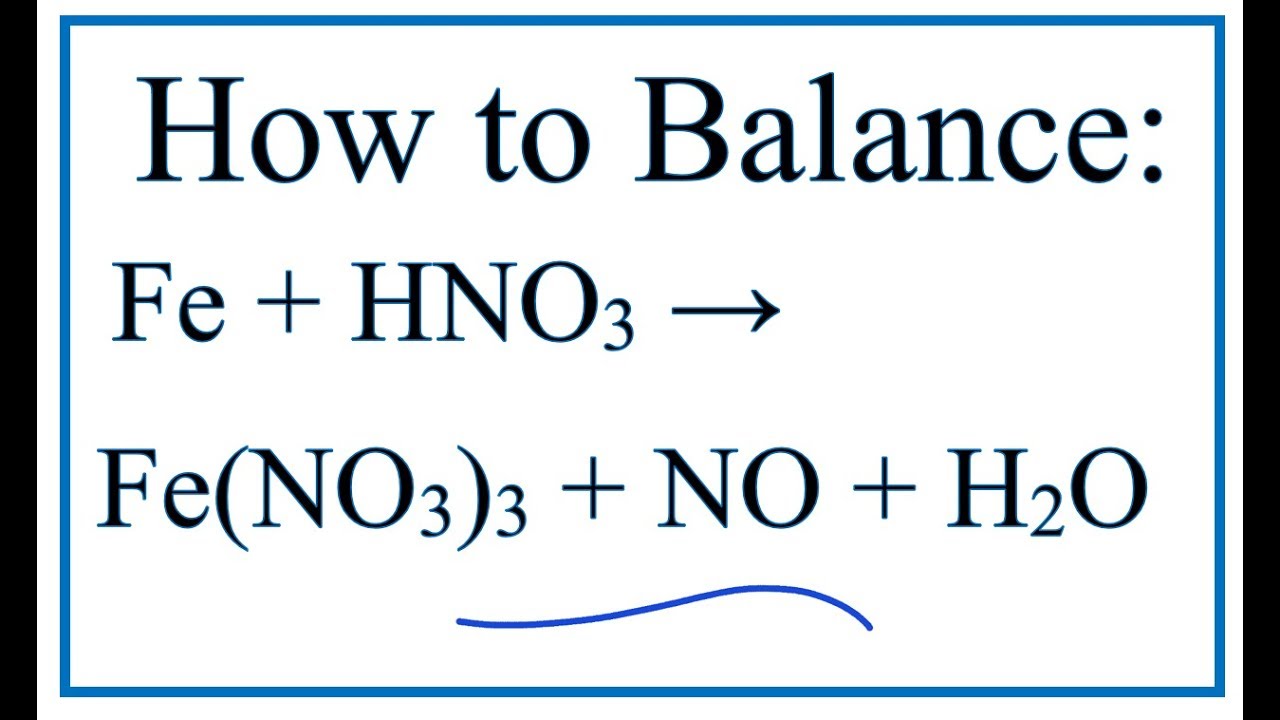
Chemical PropertiesĬhromium nitrate is used in the manufacture of alkali metal-free catalysts and in preservation. Chromium nitrate is a crystalline substance, variously stated to be green brown or purple and existing in various hydrated forms.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
